Elium For Pharma
The Knowledge-Sharing tool that helps reduce brain loss and boost R&D capabilities.
Create one central knowledge hub for everyone in your company, from clinical scientists to business professionals.
Reduce the risk of getting unnecessary industry fines.
Facilitate innovation and go-to-market capabilities.
Reduce time spent searching for information.
Drive collaboration across technical and commercial teams.
Industry Regulations and Policies
The Pharma industry is highly regulated. Offer teams up-to-date information on the key policies they need to follow.
Create one central source of truth for all industry rules that need to be followed
Group them in dedicated Spaces. Use tags to enable your teams to find the right info in a matter of seconds.
Give documents an expiry date
Industry regulations become obsolete after some time. Set an expiry date and be notified to update them.
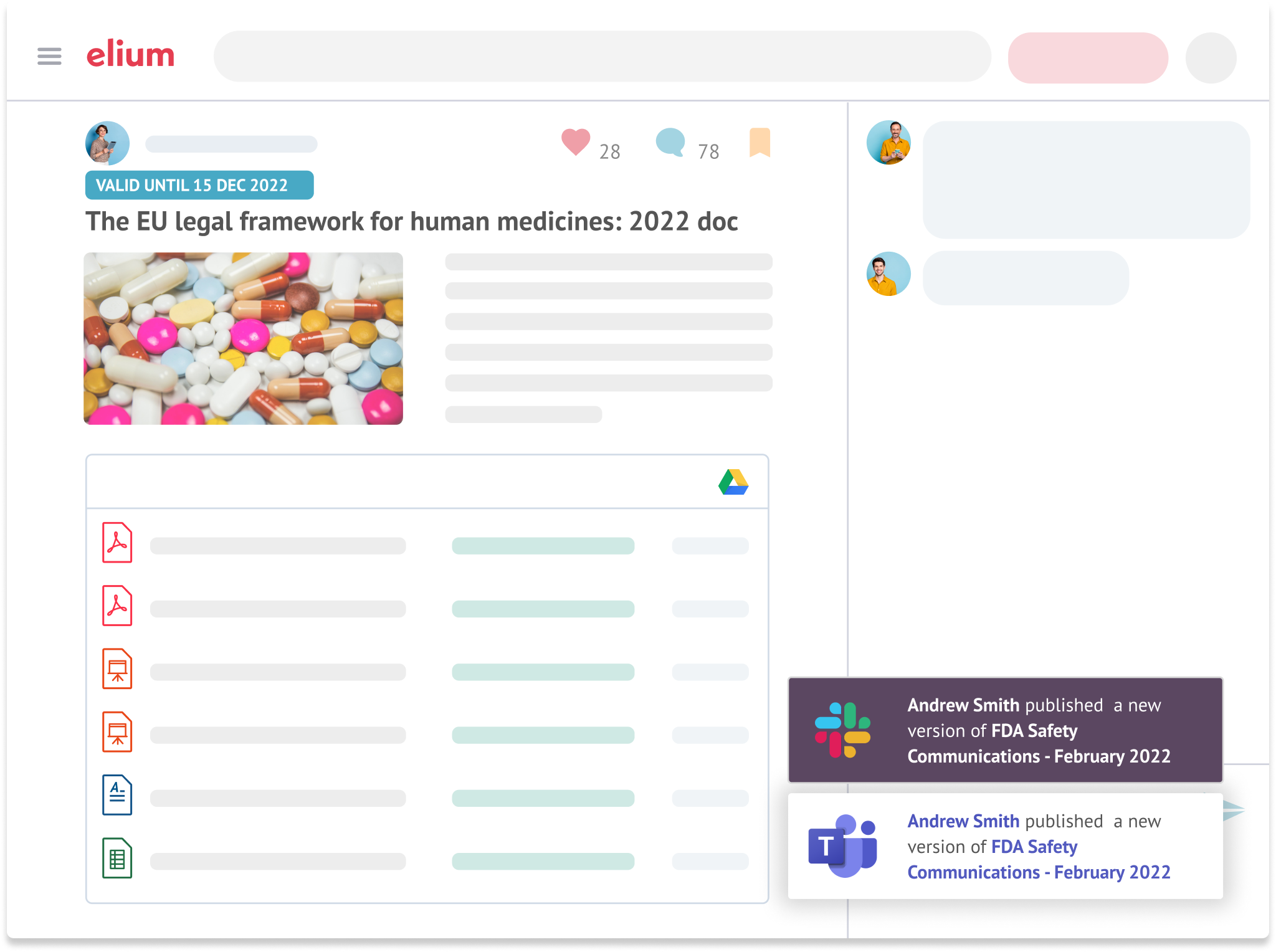
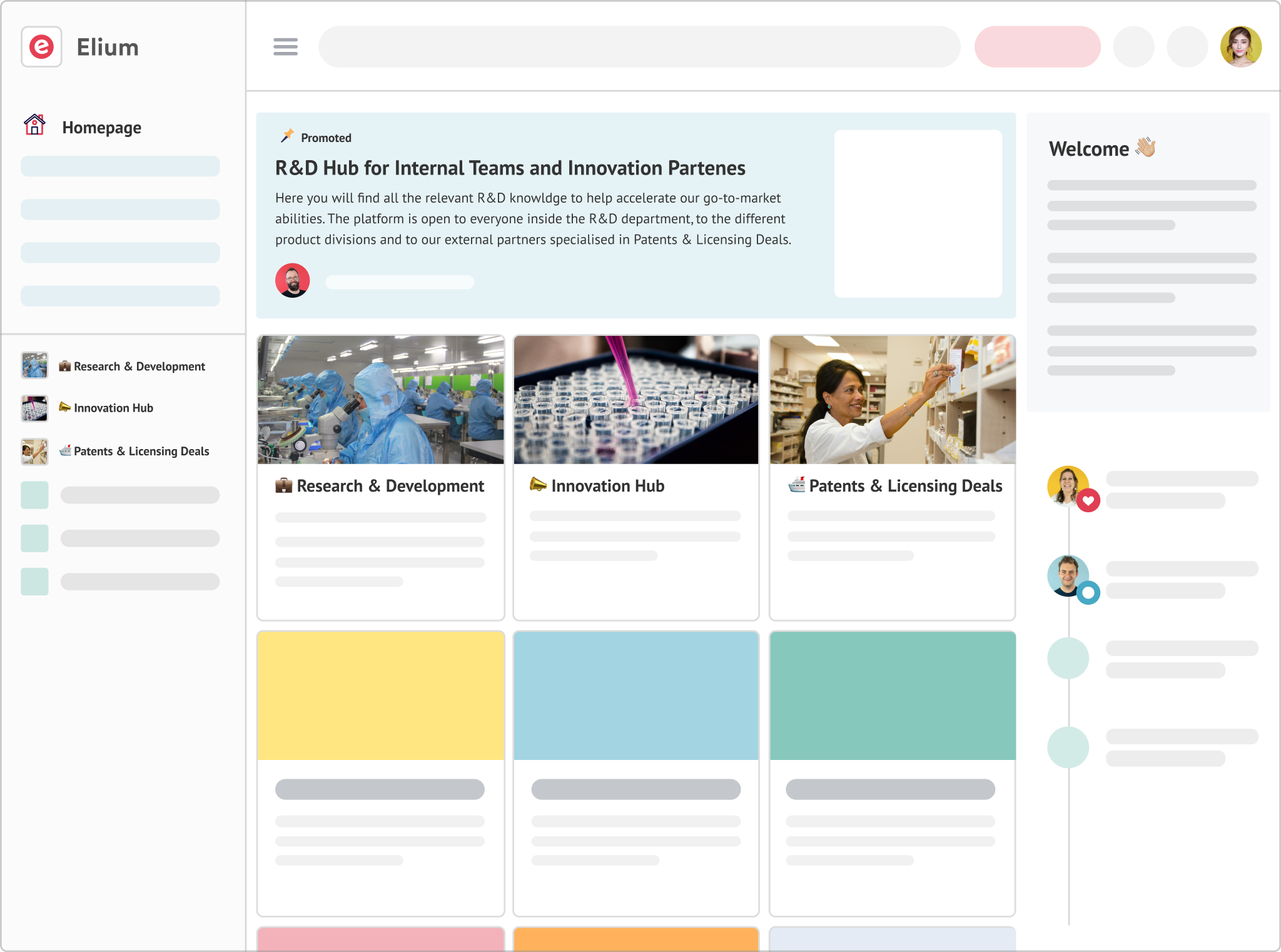
R&D and Go-to-Market Support
Long time to market is a big challenge in Pharma. Help shorten R&D cycles with instant access to crucial knowledge.
Establish Elium internally as the central hub for all R&D relevant knowledge inside the company.
Allow different departments to share data and information in a centralised manner.
Facilitate collaboration internally and with partners, around key innovation knowledge.
Seamlessly manage access levels and allow knowledge sharing with external agencies and partners on licensing deals or M&As.
Collective Intelligence and Knowledge Transfer
The Boomer Brain Drain is real. Enable experts to easily document every clinical process. Reduce intellectual capital loss.
Gather all your experts in Elium. Allow them to easily share their learnings and insights.
Create templates for all common Pharma operations such as After Action Review of clinical trials.
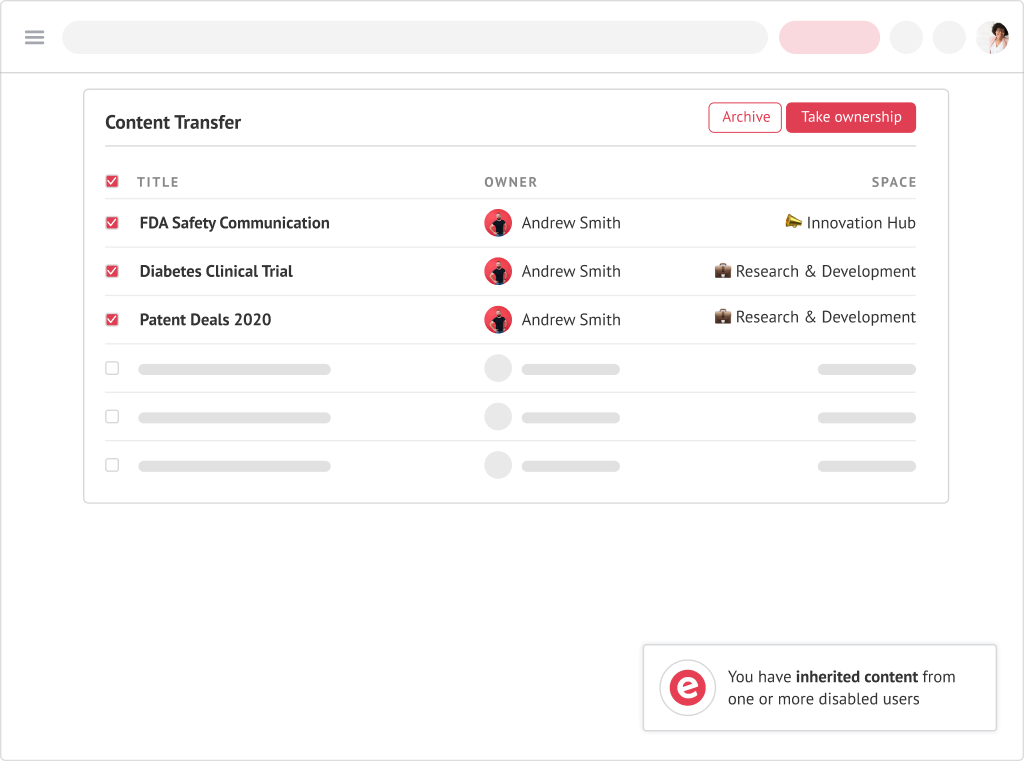
Ensure the transfer of knowledge in just one click.
With Elium’s content transfer, whenever someone leaves your company, the knowledge they created can be automatically transferred to a new owner.

Pharmaceutical Commercial Excellence
Knowledge Management does not stop with the drug development process. Connect the sales staff with physicists.
Allow for Commercial and Technical Teams to seamlessly share trusted knowledge in Elium.
Everything from product technical specifications, to sales pitches, can be centralised and shared in Elium. The knowledge is available via the mobile app too.
Facilitate collaboration and feedback giving.
With the available collaborations options (comments, annotations), encourage a reliable feedback loop from commercial staff, who are directly in link with clients.
In a nutshell, Elium empowers pharmaceutical companies to manage knowledge easily, due to the available:
Pre-defined Sharing Templates
that allows for knowledge to be shared in a structured and consistent manner.
Multi-format Sharing Options
that enable users to share text, videos, all types of files and any type of rich media.
Fully-Indexed Search Capabilities
that helps users find the good information quickly, even inside attached files.
Expiration Date and Document Versioning
to know at all times what info is up to date and see the history of changes.
Content Transfer from one user to the other
when someone decides to leave the organisation.
elium
Ready to start?
Talk to someone in our team to discover more about Elium's capabilities.
Book a demo Start a free trialFrequently asked questions
Elium lets teams create a central source of truth for all industry rules and policies, grouped in dedicated Spaces with tags for fast retrieval. Documents can be given an expiry date, and owners are notified when regulations need to be updated.
Yes, Elium can be established as the central hub for all R&D relevant knowledge inside a pharma organisation. It provides instant access to crucial information, which helps shorten R&D cycles and supports faster go-to-market capabilities.
The pharma industry is highly regulated and competitive, with thousands of micro-interactions behind every phase of drug development. Without a structured knowledge sharing platform, critical information can easily be lost, increasing the risk of fines and slowing innovation.
Yes, Elium is designed to create one central knowledge hub for everyone in a pharma company, from clinical scientists to business professionals. It drives collaboration across technical and commercial teams by making shared knowledge easy to find and use.
By offering teams up-to-date information on the key policies they need to follow — with expiry dates and notifications — Elium helps organisations stay informed and compliant. This reduces the risk of non-compliance penalties that can be costly in highly regulated industries.